site search by freefind
COVID-19
Updated: Aug 2, 2021
Update Aug 2, 2021: The rev 6 treatment has now been released to our clinic therapists for use with clients. Unfortunately, this process currently does not work on clients who need oxygen (hypoxia). Note that this treatment is NOT a substitute for vaccination - get fully vaccinated as soon as you can! (For more on this topic, we refer you to our research blog.) We continue to test treatments for the hypoxia problem.
To contact us for treatment, click on the 'Contact us' button below.
- Do you know someone with COVID-19?
We're looking for currently ill COVID-19 patients who have hypoxia (lack of oxygen in the blood) to volunteer to test our non-drug, psycho-immunology approach. The informed consent and liability form can be downloaded from here; in English; in French; in German; in Spanish; in Polish; in Russian. If you are interested in participating, please contact us by phoning (+45) 3117 5201 or by clicking on the contact button above.
A psycho-immunology approach
For readers who are not familiar with psycho-immunology, it has several unique features. Theoretically, a treatment using this approach should allow us to both (1) eliminate any of the symptoms (and pathogens) currently in an infected person, as well as (2) make people immune to getting the disease in the future. The most important feature is that symptom relief is immediate and occurs in the office during treatment, allowing one to have immediate confidence in the technique. This webpage explains psycho-immunology in more detail.
But to develop a treatment, we first need people with the actual disease who are willing to be involved in the long, difficult investigative phase. From a research perspective, we were very fortunate in that one of our research staff contracted COVID-19 (based on matching symptoms) early in the pandemic, allowing us on March 4 2020 to start investigating the disease using our new, third-generation psycho-immunological approach.
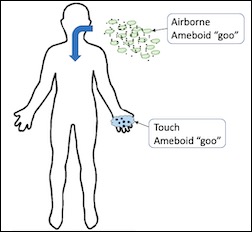
Amebic Carriers
To our great surprise, so far we haven't seen the coronavirus directly infecting humans. Instead, we see two different carriers for the coronavirus. Both appear to be ameba (both have a nucleus) and feel 'gooey' to the regressed patient. Each species infects the person quite differently:
- Airborne ameba: This pathogen floats in the air for a time after an infected person breathes them out. We cannot be sure without electrical engineering equipment, but it appears to stay airborne for a time by using an electrostatic field. If so, this suggests that (1) an infected person could be identified by using cheap sensitive electrostatic detectors, and (2) it might be possible to 'drag' the pathogen out of the air by using an electrostatic field, again a cheap and readily purchased item.
- Touch ameba: This pathogen sticks to the skin, and is passed to another person through skin contact (for example, by shaking another person's hand). It is also likely that this ameba would stick to and survive on other surfaces for a time, but we were not able to test this.
After being carried into the body via the ameba, the virus infects cells in the body. In our small sample size, the virus shows up primarily in tissue of the lower lungs.
This coronavirus is also associated with several psycho-biology markers. During the worst phase of the infection, some people can sense a sort of hungry, scary presence in space on their right side. Some people experience a sort of balloon in their upper head, from throat to eyes. And some feel that their lungs are compressed, or squeezed, and filled with water, like a barrel. Also, a psychological issue that arises for many people during symptoms can be summed up with the phrase 'I don't care', along with a strong resistance to letting go of the virus - making it harder to have them do the treatment on themselves. This feeling appears to be both a generational issue; as well as involving an active bacterial infection (prenatally acquired) that also carried a viral payload.
Virulence
This tri-pathogen model would suggest a roughly similar global distributions of pandemic virulence, which does not appear to be the case. Thus, we continue to look for possible co-factors, such as environmental toxicity or other pathogens.
Testing and validation
A timeline of our psycho-immunology research on covid-19 was presented at the 2nd International Psycho-immunology & Psychobiology Research Symposium (April 2021) via Zoom:
April 15, 2020: Given that there are at least three infectious pathogens to COVID-19, we derived a separate, simple treatment for each pathogen type. To our surprise, first treating only the viral load in a very sick client did not immediately eliminate their symptoms. However, first treating only the touch ameba in another client (which was the dominant pathogen in this person) immediately got rid of their major symptoms of breathing difficulty, exhaustion, and inability to sleep. Thus, it appears their major symptoms were due to their amebic load and not directly due to the virus.
May 3 2020: We started Phase 1 testing our rev 1.2 psycho-immunology treatments in a healthy test group to look for any potential side-effects and to estimate treatment times. Testing ended May 17. Fortunately, we did not encounter any safety issues or side effects, either in the group that was unsuccessful in using the process, or in the group that was successful. (Success was defined as the occurrence of a distinct kinesthetic experience in the volunteer that indicates the process was finished.) Our results were:
- Airborne ameba: success rate = 65%; treatment time = 89 minutes with SD = 42 minutes; N = 23.
- Touch ameba: success rate = 84%, treatment time = 58 minutes with SD = 26 minutes, N = 19.
- Virus: success rate = 76%, treatment time = 80 minutes with SD = 53 minutes, N = 21.
May 27 2020: Today we started working on a simpler and more effective process (rev 3.0) to eliminate all three pathogens simultaneously; and on June 7, we tested it on 16 healthy volunteers. The testing revealed no safety issues or side-effects.
- New single treatment protocol: success rate: 82%; treatment time = 41 minutes with SD = 13 minutes; N = 17.
June 12, 2020: We started Phase 1b testing the rev 3.0 process on an athlete volunteer recovering from COVID-19 who still had some post-infection breathing difficulty. Our new process simultaneously eliminated both ameba types and the virus in the patient, and returned their breathing to normal. This matched our model. Obviously, this is only one case, and we will now wait to see that the changes are stable.
June 20, 2020: We are now beginning testing on currently symptomatic and post-recovery volunteers. If you have or had COVID-19 and are interested in participating in these tests, contact us by clicking on the contact button at the top of this webpage.
Sept 26, 2020: Testing revision 3.0 treatment on two hospitalized covid-positive volunteers did not eliminate their symptoms - yet the treatment worked successfully on our post-covid volunteers. We eventually identified another pathogen, a bacteria attached to the outside of the amebic carriers of the virus, that was contributing to symptoms and symptom retention. We also identified a prion-like infection held in the core of the coronavirus which was released when the viral coating was dissolved.
Thus, this difference between post-covid and active cases results was due to the presence of the non-viral pathogens in the active cases - the post-covid patients' immune systems had eliminated these non-viral pathogens, leaving only the virus. We redesigned the treatment to revision 4.0 to also eliminate all of the initial infection pathogens as well as the virus. Testing on one covid positive volunteer (N=1) immediately eliminated all their covid symptoms.
Nov 15, 2020: Our rev 4.1 treatment worked very well on our last two seriously ill covid-positive volunteers, relieving symptoms immediately at the end of the treatment session. Also, we are now seeing that our current covid volunteers have different, slightly mutated forms of the coronavirus; fortunately, the rev 4.x approach automatically covers the current range of viral strains (mutations) that we've encountered so far. Update: the next two covid positive volunteers did not respond to the treatment. We are now working on why this did not work.
Feb 7, 2021: We have modified our treatment to rev 5.0. This appears to successfully eliminate all the various co-pathogens associated with coronavirus that we've now seen, as well as mutated forms of the virus. We've now tested this rev 5.0 successfully on two covid positive volunteers (n=2). We will continue to test as we get more volunteers. Treatment time to full symptom elimination was 1.5 hrs in the first volunteer (1hr, then 0.5hr the next day) and 4 hours in the second (2hrs then 2hrs the next day). For example, loss of taste and smell fully returned during their first treatment. We are now working to find ways to speed up treatment time.
March 7, 2021: The current rev 5.0 has now been used successfully on 12 covid-positive volunteers (N=12). We are fairly confident that this approach works well for eliminating all symptoms (and by implication, the viral and associated pathogens). These includes fever/chills, cough, shortness of breath, nasal/chest congestion, fatigue, headache, muscle and body ache, sore throat, loss of taste and smell, nausea, and diarrhea. Treatment time was from 2 to 4 hours (m=3 hrs). However, we've tested on three volunteers on respirators (N=3), none of which responded to the rev 5.0 treatment. We now believe that in these more severe breathing cases, the covid triggers a lung pathogen or disease process that is not present in most covid infections. We are now testing a treatment for this specific problem.
April 29, 2021: We are now testing a rev 6.0 process. This rev 6 heals the same developmental event as in rev 5, but with a simpler, faster procedure.
August 2, 2021: We've released our rev 6 process to our clinic therapists to be used with covid clients. Note that this process does not work on clients who need oxygen.
We continue to look for more patients with active hypoxia symptoms to test new treatments.
Suggested Reading
- "Please get covid vaccinated!", PeakStates research blog, Oct 3, 2021.
- "Using Psycho-immunology for COVID-19", PeakStates newsletter #19, Aug 3, 2020.
- "Editorial: Amoebae as Host Models to Study the Interaction With Pathogens", Frontiers in Cellular and Infection Microbiology, March 19, 2019.
- "How does coronavirus kill? Clinicians trace a ferocious rampage through the body, from brain to toes". Science magazine, April 17, 2020.
- "Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1", New England Journal of Medicine, April 16, 2020.
- "Epigenetics, psychoneuroimmunology, and subcellular psychobiology", PeakStates research blog, May 3, 2016.
- "How long does the coronavirus last inside the body?", National Geographic, June 3, 2020.
- "Viable SARS-CoV-2 in the air of a hospital room with COVID-19 patients", Lednicky et al., MedRxiv Aug 3, 2020. "A Smoking Gun’: Infectious Coronavirus Retrieved From Hospital Air", New York Times Aug 11, 2020.
- "Scientists Uncover Biological Signatures of the Worst Covid-19 Cases", The New York Times, Aug 5, 2020; "Researchers find three distinct immune responses for sicker COVID-19 patients", Medical XPress, July 15, 2020; Longitudinal analyses reveal immunological misfiring in severe COVID-19", Nature, July 27, 2020.
- The Vaccine Makers Project, Excellent educational videos and lessons on vaccines grades 1-12.
Revision History
Aug 2, 2021: Released rev 6 for use in our clinics.
April 29, 2021: Testing revision 6.0 now. The proceedure is simpler and faster than rev 5, but works on the same developmental events.
Mar 7 2021: Reported more successful results with rev 5.0 process, but the treatment is not working on patients in respirators.
Feb 7, 2021: Reported on the results with rev 5.0 process.
Nov 15, 2020: Reported initial successful results for the rev 4.1 technique. We are now seeing different coronavirus strains.
Sept 26, 2020: Updated with preliminary results from active COVID-19 patients. We note there is initially a bacterial component to COVID.
Aug 2, 2020: Added the Phase 1b effectiveness trial disclosure and liability PDF.
June 20, 2020: Start testing of COVID-19 volunteers.
June 16: Added the results of an improved process test.
May 29, 2020: Added the first psycho-immunology process test results.
May 4, 2020: First webpage on our coronavirus psycho-immunology research and treatment.
...or visit our Forum

